Hydrogen is an indispensable carbon-neutral energy carrier, and its large-scale practical application mainly depends on two key factors: the safety of storage technologies and the enhancement of hydrogen storage density. Solid-state hydrogen storage using magnesium hydride (MgH2) exhibits enhanced safety and high gravimetric density (7.6 wt.%). However, its practical deployment remains constrained by its sluggish de/hydrogenation kinetics, requiring considerably high activation energies.
Recently, a collaborative team led by Associate Professor Huang Jianmei and Professor Yan Xiaojun from the School of Energy and Power Engineering at Beihang University, together with Professor Shui Jianglan from the School of Materials Science and Engineering, reported a Ni/Co heteronuclear dual-atom catalyst supported on a TiO2 carrier, achieving efficient bidirectional catalysis in the MgH2/Mg hydrogen storage system. The related findings were published in Nature Communications under the title "Bidirectional catalysts with dual-atom dynamic d-band centre modulation and support self-reconstruction for de/hydrogenation in MgH₂/Mg."

The first authors of the paper are Jin Jinlong, a 2023 Ph.D. student, and Zhang Jiyue, a 2022 master's student, both from the School of Energy and Power Engineering at Beihang University. The corresponding author is Associate Professor Huang Jianmei from the School of Energy and Power Engineering, and the primary affiliation is the School of Energy and Power Engineering, Beihang University.
The researchers rationally design and construct a heteronuclear dual-atom catalyst, Ni1Co1@TiO2. In this system, Ni and Co bidirectionally modulate the d-band centres, enabling synergistic and complementary catalysis. Specifically, Ni serves as the primary active site for Mg–H bond cleavage during dehydrogenation, facilitated by the Co-induced d-band centre downshift. Conversely, Co acts as the primary active site for H2 dissociation during hydrogenation via the Ni-triggered d-band centre upshift. Simultaneously, self-reconstruction of titanium species and oxygen vacancies (OV), coupled with strong metal-support interactions (Ni/Co–TiO2), accelerates interfacial electron transfer and inhibits metal atom migration. This synergy significantly enhances both reaction kinetics and cycling stability, showing great promise for large-scale hydrogen-storage applications.
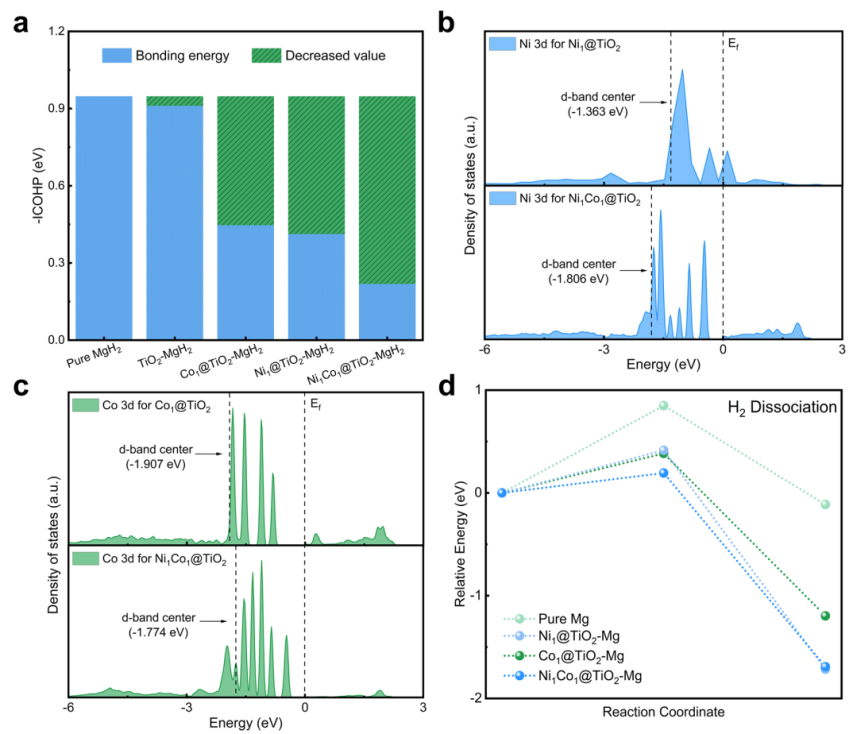
Fig. 1 Theoretical calculations for Ni1Co1@TiO2-MgH2/Mg
The ball-milled Ni1Co1@TiO2-MgH2 composite achieved a low initial dehydrogenation temperature (168 °C), with an accelerated initial hydrogen desorption rate of 3.02 wt.%·min−1at 275 °C, while enabling efficient room-temperature hydrogen absorption (3.79 wt.% within 30 min at 30 °C). The system exhibited outstanding kinetics, showing low Ea for desorption and absorption, and maintained 97.6% capacity retention over 100 de/hydrogenation cycles. This work enhanced hydrogen storage performance in MgH2-based systems while proposing a universal design framework for reversible chemical processes.
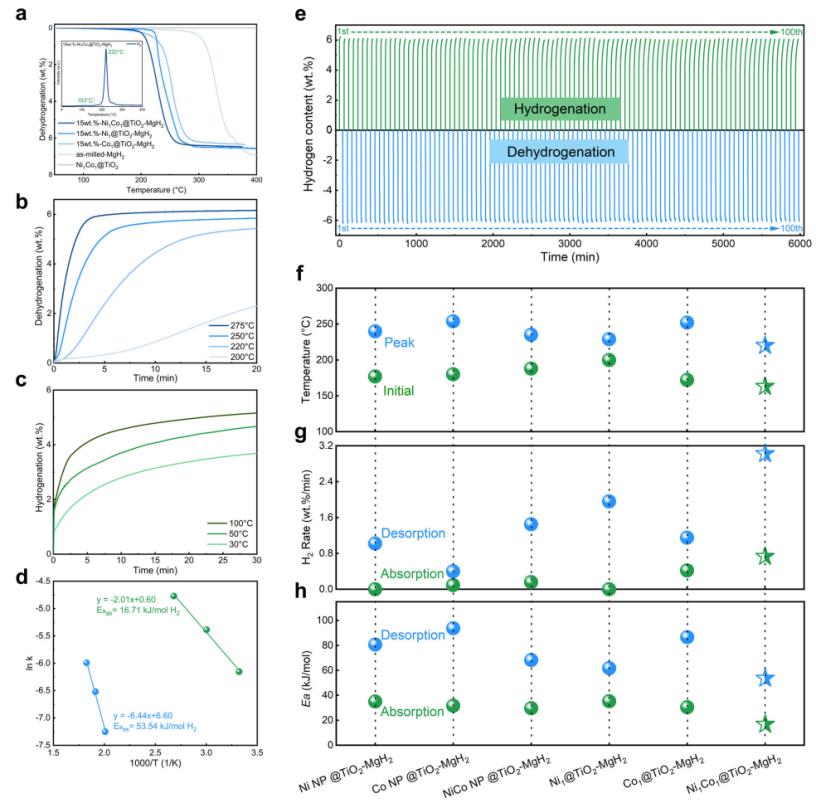
Fig.2 Hydrogen storage performance of 15 wt.%-Ni1Co1@TiO2–MgH2
Furthermore, this study systematically reveals the dynamic evolution of the TiO2 support during hydrogen absorption/desorption cycles. The self-reconstruction of Tix+ and OV within the TiO2 support and strong metal-support interactions (Ni/Co–TiO2) effectively promote interfacial electron transfer and suppress metal atom migration, remarkably enhancing reaction kinetics and cycling stability.
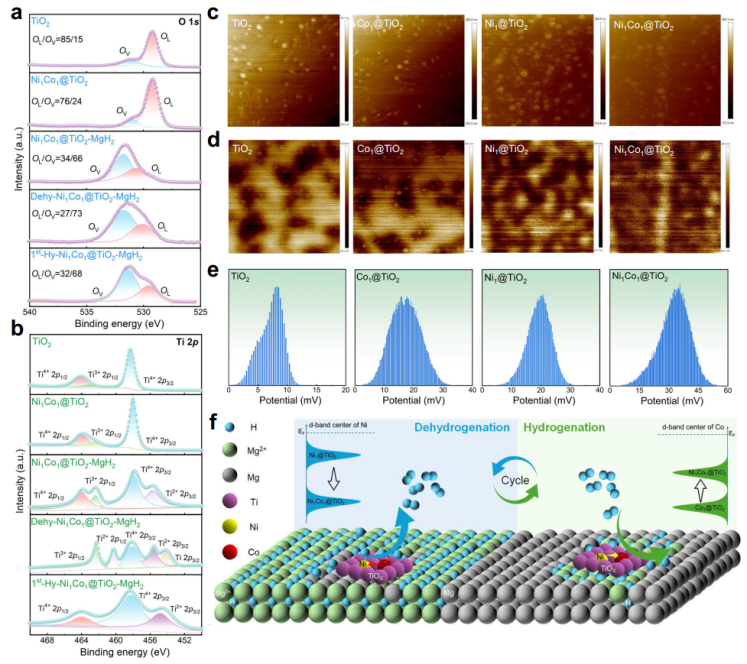
Fig.3 Synergistic mechanism of Ni1Co1@TiO2
The synergistic strategy of "dual-atom dynamic d-band centre modulation" and "support self-reconstruction" proposed in this work offers a new approach to overcoming the kinetic bottlenecks of reversible hydrogen storage reactions. This atomic-level structural elucidation provides a fundamental basis for understanding the enhanced catalytic properties and exploring the potential applications of this dual-atom catalyst system.The catalytically enhanced MgH2 material developed in this study advances the development of solid-state hydrogen storage and promotes the large-scale application of hydrogen energy.
Link to the article: https://www.nature.com/articles/s41467-026-70604-y
Editor: Liu Tingting

 Beihang NewsSpring tree-planting brings greenery to Beihang campusMore
Beihang NewsSpring tree-planting brings greenery to Beihang campusMore Beihang NewsBeihang University signs cooperation agreement with Yonsei UniversityMore
Beihang NewsBeihang University signs cooperation agreement with Yonsei UniversityMore Beihang NewsBeihang Retired Faculty Choir debuts on Beijing Music Radio to celebrate Space DayMore
Beihang NewsBeihang Retired Faculty Choir debuts on Beijing Music Radio to celebrate Space DayMore Beihang NewsWatch: How are Beihang University students pursuing sustainable development?More
Beihang NewsWatch: How are Beihang University students pursuing sustainable development?More Beihang NewsSecretary General of T.I.M.E. Association visits BeihangMore
Beihang NewsSecretary General of T.I.M.E. Association visits BeihangMore Beihang NewsFaculty spotlight: Guillaume Merle's 13 years bridging Sino-French educationMore
Beihang NewsFaculty spotlight: Guillaume Merle's 13 years bridging Sino-French educationMore